|
A good example of this can be seen on a ternary (3 component) chemographic projection if the phases of interest plot in a co-linear fashion (i.e. In some cases, a given mineral assemblage may be represented by a sub-set of the whole system if fewer components are needed to define the compositions of the observed mineral assemblage-this is known as a degenerate system. binary systems such as carbonates, CaCO 3 - MgCO 3 alkali feldspars, NaAlSi 3O 8 - KAlSi 3O 8, etc.). In some geologic systems it is convenient to define the components in terms of end-member compositions (e.g. 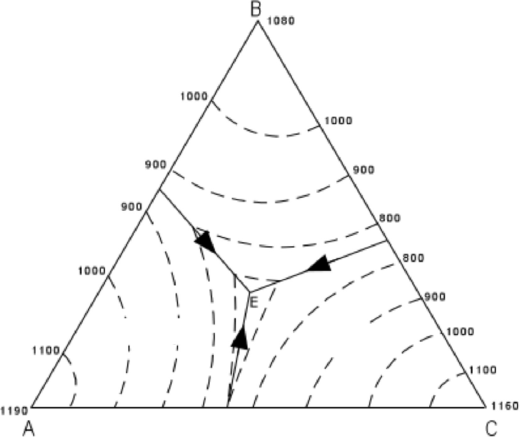
However, because Ca and Mg are in a 1:1 ratio in both dolomite and diopside (and not present in quartz or carbon dioxide), we can consider this a ternary system with components: CaMgO 2, SiO 2, and CO 2 Normally we would pick the 4 components: CaO, MgO, SiO 2, and CO 2 based on the simple oxides. Consider the reaction:ĬaMg(CO 3) 2 + 2 SiO 2 = CaMgSi 2O 6 + 2 CO 2ĭolomite + 2 Quartz = Diopside + 2 Carbon Dioxide If two possible components always occur in the same proportions in multiple phases in a system, these can be combined into a single component (remember, we are always trying to define the minimum number of components required to make all the phases in the system). C is the minimum number of chemical components required to constitute all the phases in the system For historical reasons, geologists normally define components in terms of the simple oxides (e.g. solid mineral assemblages, immiscible silicate and sulfide melts, immiscible liquids such as water and hydrocarbons, etc.) Phases may either be pure compounds or mixtures such as solid or aqueous solutions-but they must "behave" as a coherent substance with fixed chemical and physical properties. 
It is possible to have two or more phases in the same state of matter (e.g. Every unique mineral is a phase (including polymorphs) igneous melts, liquids (aqueous solutions), and vapor are also considered unique phases. P is the number of phases in the system A phase is any physically separable material in the system. Gibbs Phase Rule is expressed by the simple formulation: A solid understanding of Gibbs' Phase Rule is required to successfully master the applications of heterogeneous phase equilibria presented in this module. The reaction curves actually represent the condition (or the locus of points in P-T space) where ΔG rxn =0 for more information on this point see Gibbs Free Energy. In the simplest understanding of phase diagrams, stable phase (mineral) assemblages are represented as "fields" (see colored areas on the figure to the right) in "P-T space", and the boundaries between stable phase assemblages are defined by lines (or curves) that represent reactions between the phase assemblages. Gibbs' Phase Rule also allows us to construct phase diagrams to represent and interpret phase equilibria in heterogeneous geologic systems. Gibbs' Phase Rule provides the theoretical foundation, based in thermodynamics, for characterizing the chemical state of a (geologic) system, and predicting the equilibrium relations of the phases (minerals, melts, liquids, vapors) present as a function of physical conditions such as pressure and temperature. Use of minerals in geochronology and thermochronology.minerals, melts and/or fluids) to characterize and quantify petrogenetic processes and 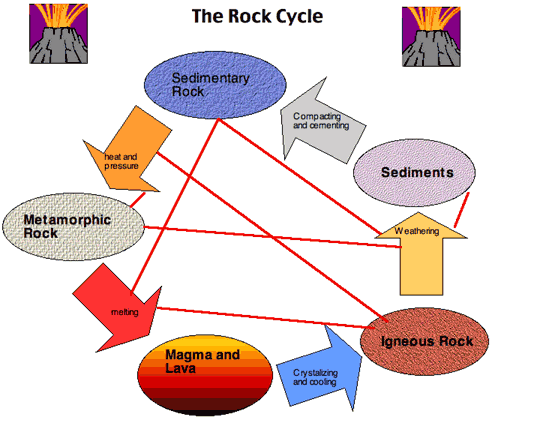
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
